IoT brace
Smart sensor for monitoring of scoliosis treatment braces
The challenge
Each year about 2% of worldwide teenagers suffer of scoliosis: from moderate to severe scoliosis must be treated with braces (spinal orthosis) to counteract the evolution of scoliotic curve by applying a mechanical stress against the body and favouring the correct posture of rachis.
Despite the need of recording the exerted pressure expressed by orthopaedic specialists, there is currently no device available, in the market, able to quantitatively measure acting forces inside the braces and to monitor the wearing time.
Market analysis outlines big potential in the scoliosis management market, stressing increasing year to year demand still not satisfied by products on the market. It also identifies among main drivers the advancement of technology with tracking devices that can monitor the patient health evolution.
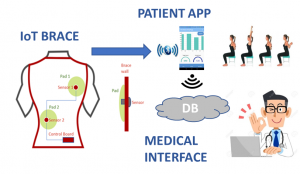
Protolab aims at:
- developing an innovative plug & play wearable sensor kit for monitoring the pressure inside scoliosis braces,
- improving the compliance with medical prescriptions,
- boosting the patient’s active participation to reach satisfying results from the care treatment.
Digitization can boost the development of automated analysis to frequently check treatment progress, while the availability of gathered data can be used by predictive algorithms to favour rapid intervention in case of wrong trends, thus favouring the active participation of the patient to the care treatment.
Product development and FED4SAE support
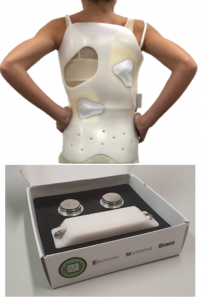
Leveraging the collaboration with CEA-Leti, STMicroelectronics, and IRT Nanoelec, Protolab developed a prototype system based on STM32WB microcontroller integrating all the hardware means to support BLE 5.0 and IEEE802.15.4 wireless standards (e.g., ZigBee 3.0, Openthread). The STM32WB is used to collect data from sensors, to process, and then to transfer them via BLE to a dedicated smartphone App.
Tests of the new prototype data communication under realistic conditions were performed in collaboration with Products & Technologies Living-Lab (PTL) of IRT Nanoelec at CEA-Leti in Grenoble. The cybersecurity and privacy risk analysis and the evaluation of regulatory constraints related to patient’s data management were assessed as well with the support of CEA-Leti.
Blumorpho’s innovation management services helped Protolab identify the strategy and the statutory constraints to secure the go-to-market strategy as well as to seek investors.
The results
Protolab developed a miniaturized and integrated solution tested in a realistic environment.
Product qualification and certifications were launched in July 2020. Protolab planned to enter the market in early 2021 (customer acquisition).
Embrace enters a Global Scoliosis management Market estimated in 2B$. A rough estimate of investments needs, viability and sustainability of business has been carried out with an analysis of incremental costs and revenues that Protolab will tackle to enter the new market.
The estimated financial requirement is about 1.4 Mn EUR, while the Break-Even Point will be reached within five years at about 3000 kits/year.
 |
Healthcare |
Protolab srl: High tech SME developing innovative complete solutions tailored to customer needs and providing integrated systems & services.
Engaging young patients
‘Patient’s motivation plays a key role for therapy success. Our scope is to engage young patients and improve their awareness to maximise achievable results and give them back satisfaction because they feel to be protagonists in their own therapy.’
(R. Pierobon, Project Coordinator at Protolab SRL)
Impact
- Protolab started the process to obtain the support from the Italian Health System to refund IoT braces
- Protolab raised the most interest from a panel of Business Angels during the INPHO venture Event, October 2020
- Protolab was awarded the 1st prize in the Smart Anything Everywhere contest for the innovation approach to address scoliosis treatment with digital and sensors approach
Supported by



![]()
Authors and Contributors: CEA, STMicroelectronics, Digital Catapult
All images © Protolab SRL

